Overview
It is the responsibility of the Principal Investigator (PI)/Project Team to ensure correct compliance items are linked in the Award record (AWD) or Modification (MOD) (e.g., Human Subjects Applications (HUMs); Animal Protocols (PROs), etc).
Holds
Office of Research and Sponsored Projects (ORSP) will evaluate if there are any outstanding items that need to be completed prior to processing the awards. The ORSP Project Representative will place an Award on Hold (PR Hold or Manage Hold) if any of the following are missing:
- Agreement Acceptance Request (AAR)
- Regental Action Request (RAR)
- Human Subjects Application (HUM ID)* if human subjects are indicated
- Animal Protocol (PRO ID)* if vertebrate animals are indicated
- Institutional Biosafety Committee Application (IBCA ID)*
- Awaiting Dates from Sponsors (required to set Start and End dates of the project)
- Other
- e.g., Risk Mitigation Approval (if Research Security Office review of a security risk term/condition is indicated)
*ORSP will place the Award on hold if a HUM, PRO, or IBCA is required and the PI/Project Team have not yet provided the related compliance application's ID.
Award Workflow
Here is a diagram of an Award's State and progression through the workflow with routing stops: Initial Award Processing > Hold > PR Signoff > Compliance Hold > Account Assignment > Financial Processing > Pending Activation > Active.
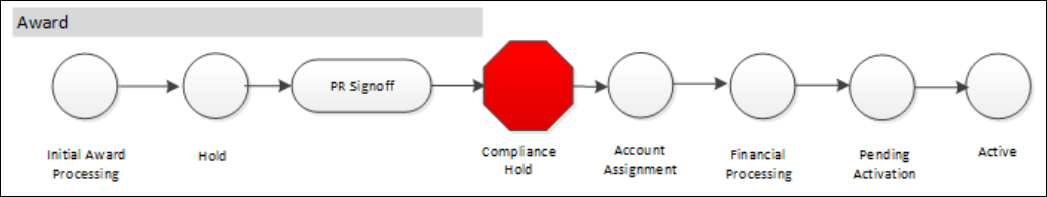
Traffic signals and colors indicate progress and where action is required. For more details see the Award Status Map Reference.
Compliance Holds
After Project Representative PR Signoff, eRPM automatically checks for required approvals and places the Award in a State of Compliance Hold if any of the following are missing:
- Institutional Review Board (IRB) approval*
- IRB Approval Future dated - HUM
- Institutional Animal Care and Use Committee (IACUC) approval
- IACUC Approval Future dated - PRO
- Institutional Biosafety Committee (IBC) approval
- IBC Approval Future dated - IBC
- Conflict of Interest Committee approval (PHS FCOI and/or PI/ORSP)
- Conflict of Interest (COI) Staff Review and AWD
- Conflict of Interest (COI) Staff Review and AWD for U-M Regental Action Request (RAR) approval
- Human Pluripotent Stem Cell Research Overright (HPSCRO) Ancillary Committee Review
- Radiation Policy Committee (RPC) Review
- Export Controls (EC) Review completion
- Research Information Security Oversight (RISO) Review Required
- Other Support Review Required
- M-Inform Disclosures Required
- Compliance Training (PEERRS) completion
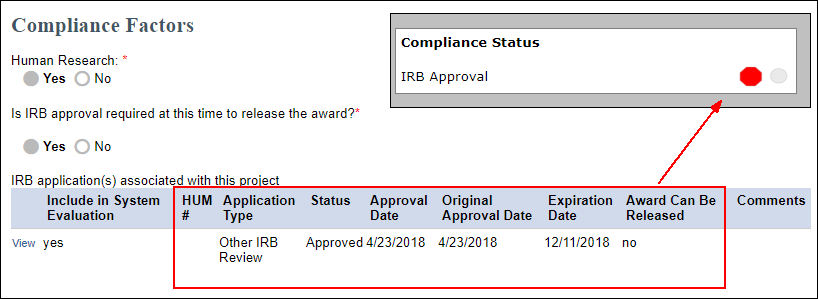
*Institutional Review Board approval status will display pending (red) for any HUM that is an Umbrella or Ceded (Other IRB Review) application type. Ceded applications will be approved by U-M's IRBs but are not considered award-able until the Project Team uploads the other IRB's approval letter in eResearch Regulatory Management (eRRM). In the example below, the Other IRB Review is Approved, but the Award can not be released. So the Compliance Status meter on the Award workspace will show pending (red).
Compliance Factors with an associated IRB Application
Once the compliance approvals are completed, the Award moves on to Office of Contract Administration (OCA) if applicable, Shared Services Center (SSC) and Sponsored Programs for Project/Grant account assignment and M-Pathways Financials set-up.
Note Updates from other eResearch systems (eRRM, eRAM, etc.) will happen in real-time; however, the check to evaluate and release a Compliance Hold will occur every 60 minutes. (i.e., Project Teams may notice a slight time delay between IRB Approval and release of compliance hold.)
Compliance Status
Within the Award Workspace, a Compliance Status meter displays to indicate an Award’s required compliance actions or approvals, as indicated on the PAF or by ORSP. Traffic signals (status signals) indicate if processing is stopped due to an outstanding compliance action or hold. The meter will only show the approvals that are applicable to the Award. Workspace messages will display directly above the Compliance Status.
Award Workspace
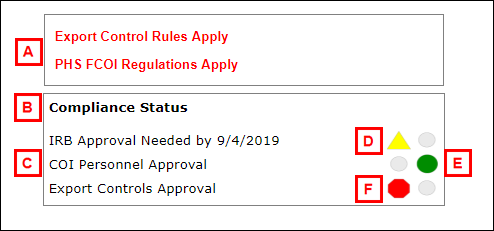
| Letter | Section/Field |
|---|---|
| A | Workspace message(s) (e.g., Export Control Rules Apply, PHS FCOI Regulations Apply) |
| B | Compliance Status meter |
| C | Compliance approvals required (e.g., IRB Approval, COI Personnel Approval, Export Controls Approval, Risk Mitigation) |
| D | Status signal- Yellow triangle (warning of future condition related to compliance, such as delayed onset of human subject work) |
| E | Status signal- Green circle (approved, complete) |
| F | Status signal- Red hexagon (pending approval, on hold) |
What can I do?
See the Manage Holds and Compliance Holds on AWDs and Mods reference page on ORSP's website for detailed guidance.
