Overview
Study Team Members (U-M and sponsored affiliates) and Core Committee Staff can use the Post Correspondence activity to communicate with others involved with the study. Post Correspondence is a communication tool only and will not update or submit your application.
Important The posted messages are permanent and visible to anyone with access to the Study Workspace (e.g., Study Team Members, Core Committee Staff, IRBs, Ancillary Committees, regulatory officials). Do not attach a document or comment with Protected Health Information (PHI).
Navigation
Role: PI/Study Team Member or Core Committee Staff > Home Workspace > Study Workspace
Step-by-Step Process
- Click the Post Correspondence activity.
- Enter or edit the Email Subject.
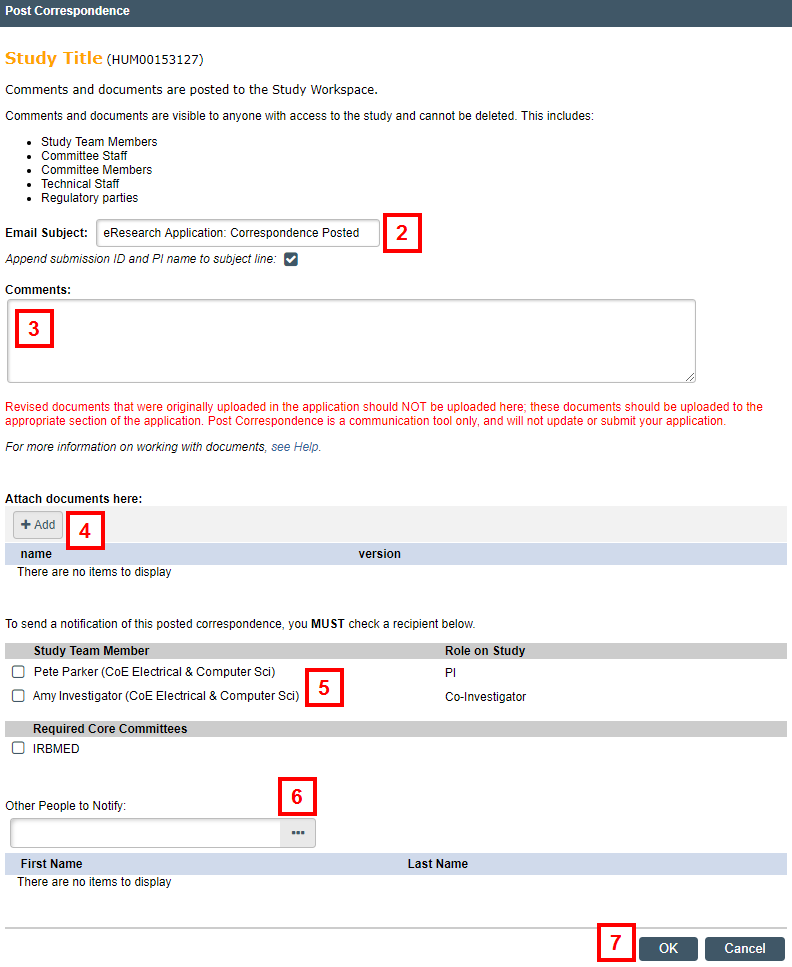
- Enter Comments.
- If applicable, click Add to attach documents.
Note Revised documents that were originally uploaded in the application should NOT be uploaded here. They should be uploaded to the appropriate section of the application. - Select recipients to notify of your correspondence.
- If you need to include non-study team members in the correspondence, click Browse
 in the Other People to Notify field and select the person from the list. Repeat the process for all additional people.
in the Other People to Notify field and select the person from the list. Repeat the process for all additional people. - Click OK.
The selected recipients will receive an email notification of the correspondence.
Note The Posted Correspondence Activity, Author, and Date display under the Activities and Correspondence section on the Study Workspace.
