Overview
After the PI/Study Team receives approval from an external (Non-UM) IRB, the Study Team must indicate approval receipt in eResearch Regulatory Management (eRRM) by uploading supporting documents.
Note Until this activity is completed, associated Awards in the eResearch Proposal Management System (eRPM) will remain in a Compliance Hold State.
HUM Study Team members with edit rights can complete the following steps on Other IRB applications to upload approval documents, which releases the associated Award's Compliance Hold State for IRB Approval in eRPM.
Navigation
Role: Study Team Member > Home Workspace
Step-by-Step Process
- Click the In Progress or Approved tab to locate the applicable Review by Other IRB Type application study.
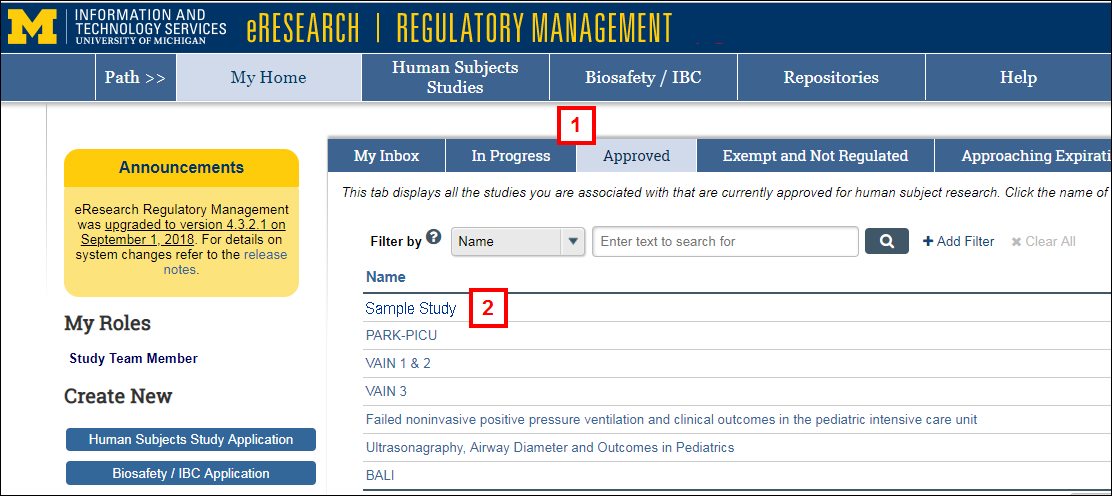
- Click the Name of the application.
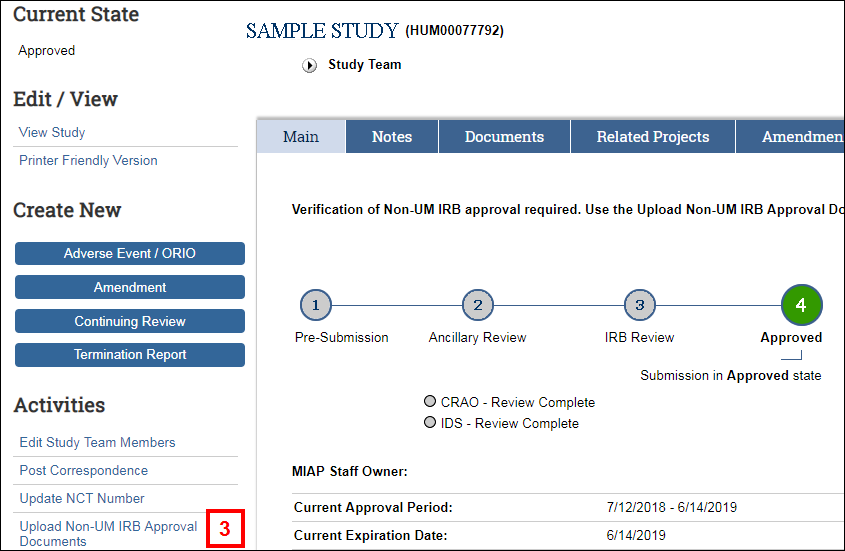
- Click the Upload Non-UM IRB Approval Documents activity.
- Check the U-M has received approval from a Non-UM IRB to conduct the research referred to in this application box.
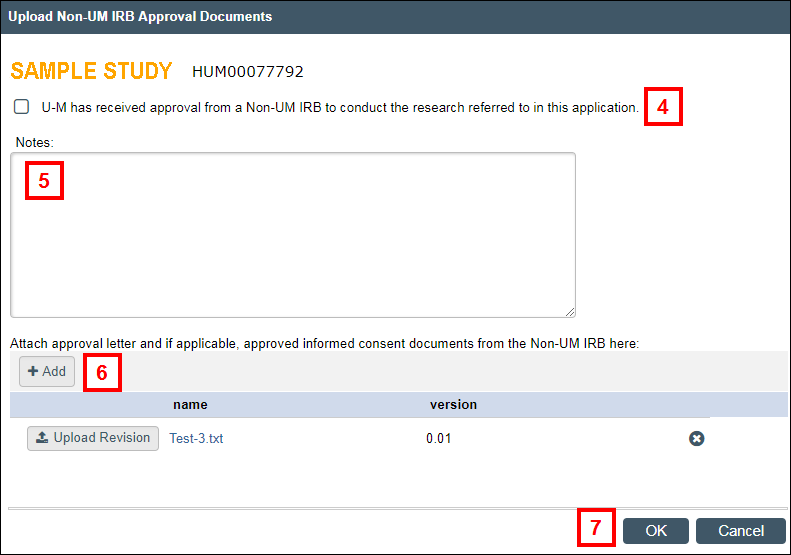
- If applicable, enter any Notes.
- If applicable, click Add to attach the approval letter and other supporting documents.
Note Click to Upload Revision or Delete existing documentation. - Click OK.
View uploaded documentation on the Documents tab of the Study Workspace under Supporting Documents.
Last Updated
Thursday, October 17, 2019
