Overview
The question "Is Continuing Review required for this application?" displays on the bottom of the activities for Expedite, Record Expedited Decision, Full Committee, and Record Committee Decision, and on the associated Reviewer Checklists (Primary, Secondary, and Regulatory Reviewers). See the examples below. This question will display on submissions that are in the states of Expedited Review, Core Committee Staff Review, Validate Expedited Decision, and Full Committee.
After determination that a CR is not required, a message will display on the Study Application Workspace: No Continuing Review Required. However, Amendments and AE/ORIOs are required when applicable. The Continuing Review button under Create New will not be available.
Navigation
Role: Core Committee Staff
Examples
If Yes is selected, then you must check the reason(s) for requiring Continuing Review.
Example of "Is Continuing Review required" question and answers
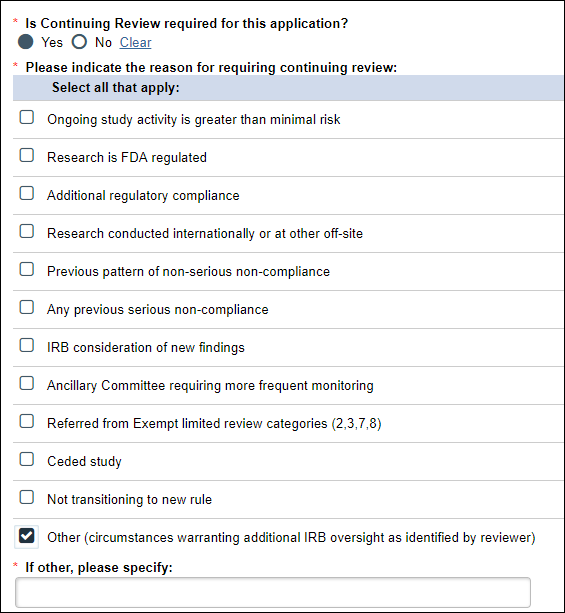
Notes
- This answer can be changed during the review process; for example, IRB staff can change the decision to either Yes or No CR required.
- Once the application is approved, an Amendment or Scheduled Continuing Review (SCR) is required to change the Continuing Review required status.
For applications in Full Committee
Record Committee Decision activity
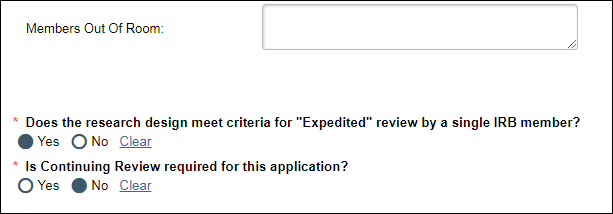
If Yes is selected for the question "Does the research design meet criteria for "Expedited" review by a single IRB member?", then the "Is Continuing Review required for this application?" question will display below.
